Essential Plant Nutrients
Share this Post
Plants require light, water, carbon dioxide, and minerals to grow. Plants are unique in that they are sessile and must receive elemental nutrients through roots in a growing medium. There are 14 elements that are essential for growth and development that plants receive through roots. Each nutrient plays a different role in the plant from structural components to regulation of plant processes. When a nutrient is not present in sufficient quantity, plant growth is negatively impacted. This paper provides a review of the role of essential and beneficial plant nutrients. Additionally, plant nutrient deficiencies and symptoms are discussed.
ROLE OF ESSENTIAL NUTRIENTS IN THE PLANT
There are 14 plant nutrients essential for proper growth and development. An element is considered essential when it fits these three criteria.
- The plant cannot complete its life cycle in the nutrient’s absence.
- The action of the nutrient is specific.
- There is no substitute for the nutrient.
The essential nutrients include six macronutrients and eight micronutrients. Macronutrients are defined as being needed in large (usually >0.1% or 1000 ppm) quantities in the plant whereas micronutrients are needed in relatively small quantities (usually >0.1 ppm). The six macronutrients are nitrogen, phosphorus, potassium, calcium, magnesium, and sulfur. The eight micronutrients include iron, manganese, zinc, boron, copper, chloride, nickel and molybdenum. The function of each plant nutrient is listed below.
MACRONUTRIENTS
Nitrogen (N) – essential component of amino acids and nucleic acids, which are the building blocks of proteins and DNA, respectively. Nitrogen is an essential element in chlorophyll used for photosynthesis, energy transfer, and carbohydrate utilization.
Phosphorus (P) – plays a role in energy transfer processes, notably through ADP and ATP. Phosphorus is a structural component of cell membranes, nucleotides (DNA and RNA), coenzymes, and phosphoproteins.
Potassium (K) – functions in enzyme activation, protein synthesis, and osmotic balance in cells. Potassium transport across cellular and organellar membranes plays a role in the energy status of plants.
Calcium (Ca) – plays a structural role in cellular membranes and cell walls. Calcium functions in N metabolism and protein formation, movement of carbohydrates and nutrients, and cell division and elongation.
Magnesium (Mg) – is a component of the chlorophyll molecule crucial to photosynthesis. Magnesium is also a cofactor for many enzymes, regulates components of cellular membranes and stabilizes proteins, DNA, and RNA
Sulfur (S) – is a structural component of amino acids in proteins and plays a role in formation of chlorophyll for photosynthesis. Additionally, sulfur is involved in enzyme activation and fatty acid synthesis.
MICRONUTRIENTS
Iron (Fe) – acts as an enzyme activator and an important component of certain proteins. Iron is essential for chlorophyll synthesis and photosynthetic reactions.
Manganese (Mn) – activates enzymes that are involved in cellular respiration and synthesis of important molecules such as amino acids, lignin, and hormones. Manganese also plays an essential role in photosynthetic reactions.
Zinc (Zn) – is an important component of enzymes serving either catalytic, coactive or structural functions. Zinc also plays a role in hormone production, chlorophyll synthesis and cellular membranes.
Copper (Cu) – activates enzymes necessary for various plant growth functions. This element is used in chlorophyll formation, lignin synthesis, and production of various other molecules such as vitamin A.
Boron (B) – is involved in root elongation and metabolism of biomolecules such as nucleic acids for DNA or RNA, amino acids and proteins, nitrogen, sugars, and starches. Boron also plays a role in flower formation, seed production, and stability of cellular membranes.
Nickel (Ni) – plays a role in enzymes involved in nitrogen metabolism. Nickel may help plant responses to pathogens.
Chlorine (Cl) – is required for activity of enzymes involved in amino acid synthesis, starch catabolism, and release of energy from ATP. Chlorine is essential for photosynthesis and osmotic regulation of plant cells.
Molybdenum (Mo) – occurs in more than 60 plant enzymes, many of which are involved in nitrogenmetabolism in the plant
BENEFICIAL PLANT NUTRIENTS
Beneficial plant nutrients are elements that assist plants with growth and development but are not essential for growth. Plants can function in the absence of beneficial plant nutrients. The beneficial plant nutrients are aluminum, cobalt, selenium, silicon, sodium, and vanadium. In general, the benefits of these nutrients to plants are often species-specific. Low levels of these elements can result in stimulatory effects while high levels often result in toxicity. Beneficial elements often help plants in dealing with biotic or abiotic stresses. Low levels of these elements in growing media or inputs will likely cause no harm to plants. On the other hand, additions of these elements during the growth cycle is not necessary to achieve target yields and quality
NUTRIENT DEFICIENCIES
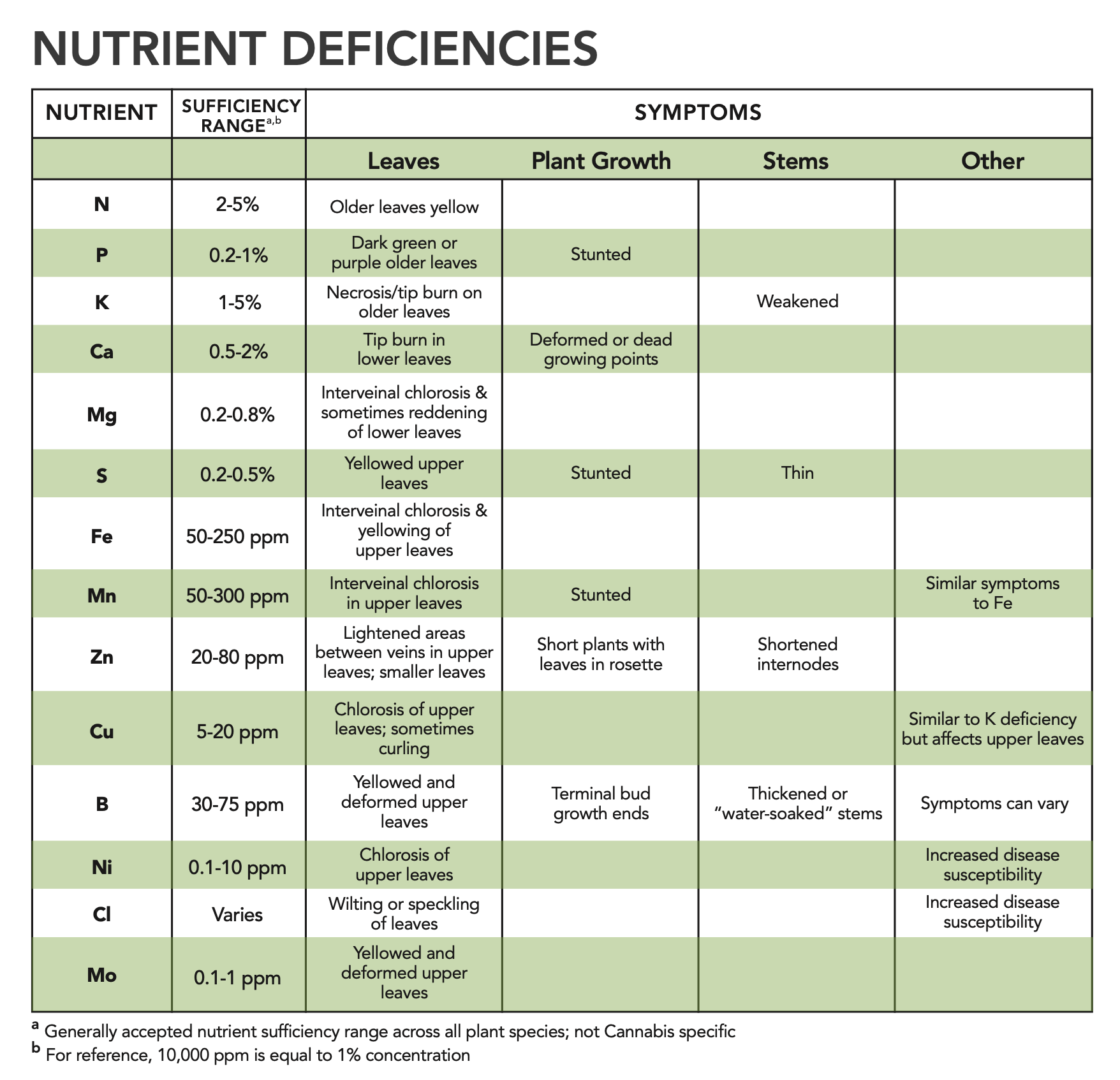
Plant nutrients often limit plant growth, resulting in deficiencies. Deficiencies in plants can occur before visual symptoms are observed. Testing of plant leaf samples is a great tool for diagnosis of nutrient deficiencies. In Cannabis, there are no established nutrient sufficiency ranges in leaf tissue. However, there are generally accepted nutrient sufficiency ranges developed in other crops that can growers can use as a guideline for Cannabis.
Visual symptoms of nutrient deficiencies usually affect leaves, plant growth, and/or stems. The
key symptoms of deficiency for each essential plant nutrient are listed in the table below. When assessing visual symptoms, upper (younger) and lower (older) leaves will often show different symptoms based on the nutrient in question. Each nutrient has different levels of mobility within plants. Mobile nutrients such as N, P, K, Mg, Ni, and Cl tend to show deficiency symptoms in lower leaves while immobile nutrients tend to show deficiency symptoms in upper leaves. Many nutrient deficiencies can look similar. Understanding the growing media and nutrient inputs can help give clues as to the nutrient deficiency in question
CONCLUSIONS
Plants rely on uptake of elements through their roots to provide nutrients essential to growth and development. Essential plant nutrients have specific functions in plants and are each needed in unique quantities. Some elements can benefit plant growth and development but are not necessary. When a plant nutrient is limiting, visual signs can give clues as to which nutrient is deficient. Nutrient uptake in plants is a complex phenomenon that is often species-specific and under the influence of conditions in the growing media as well as fertilizer inputs.