Water Quality
Share this Post
PLANT HEALTH FACTORS
Water quality is one of the most important factors affecting plant growth, as unwanted components in water can interfere with nutrient availability and uptake by the plant. When improving your grow operation, water should be one of the key areas evaluated. Even with a filter in place, problems may still arise if quality is not closely monitored. In this paper, we will discuss the steps you should take to evaluate your water, what issues might be present, factors affecting water quality and troubleshooting solutions.
FACTORS AFFECTING WATER QUALITY
Water Source: Municipal vs. Private Well: All natural water sources will contain some levels of dissolved salts and trace minerals. The source of the irrigation water can drastically change the levels of salts and minerals found in water. Excessive amounts of mineral salts found in water can have a negative effect on plant health (see images) due to nutrient antagonism (Appendix B).
Untreated well water may contain high levels of fertilizer salts as a result of runoff, sediment, and calcium from water hardness. Municipal water sources may be treated with chlorine or chloramine to kill pathogens, but this can have a detrimental effect on plant health. This water source may also have high levels of sodium or potassium as a result of water softening agents used to reduce water hardness. Heavy metals occur naturally in the environment and can be found in private and municipal water sources. Many of these have the potential to accumulate to toxic levels in soil and plant tissues.
Waterborne pathogenic organisms may exist naturally or appear as a result of fecal contamination from livestock. These include viruses, fungi and bacteria such as E. coli and salmonella. Some pathogenic organisms may not have a direct impact on plant health but can most certainly affect the humans working with plants. Some waterborne organisms do have a direct effect on plant health, such as nematodes, Pythium and Phytophthora.
- Damping off caused by Pythium fungus
- Sulfur deficiency in alfalfa
- Magnesium deficiency in grape
- Calcium deficiency in sugarbeet
- Potassium deficiency in soybeans
WATER QUALITY CHECKLIST
1. Check your pH: pH is the measure of the concentration of hydrogen (H+) ions in a solution. Water (H2O) consists of hydrogen ions and hydroxide (OH-) ions. Water with a neutral pH of 7.0 has an equal balance of hydrogen and hydroxide ions. The more H+ ions that exist, the more acidic the solution. The more OH- ions that exist, the more basic the solution. Water with a pH between 5.5 and 7.0 is an ideal range for horticulture. Once water pH increases beyond neutral, there is an increased risk for limited nutrient availability and solubility. A pH above 8.5 may also have high alkalinity or high total dissolved solids (TDS), meaning that levels of carbonates or salts are elevated, further contributing to limited nutrient uptake. Water with a pH below 5.5 may have an excess of hydrogen ions and a decreased buffering capacity.
2. Check your EC or TDS: EC is a measure of electrical conductivity in water, and TDS is a measure of total dissolved solids in a solution. EC and TDS are not the same unit of measure, but are closely related and either one can be used to determine water hardness.
Most EC meters will convert to ppm on either a 500 or a 700 scale, so it is important to note which scale your meter is converting from. If EC or TDS is above the desired range, filtration is the best option to reduce the amount of salts in the water.
Although most horticulture research recommends a TDS below 640 ppm for best results, hydroponic and soilless cannabis cultivation tends to show higher sensitivity to pH fluctuation and salt buildup when TDS is above 200 ppm (500 scale) or 0.4 EC.
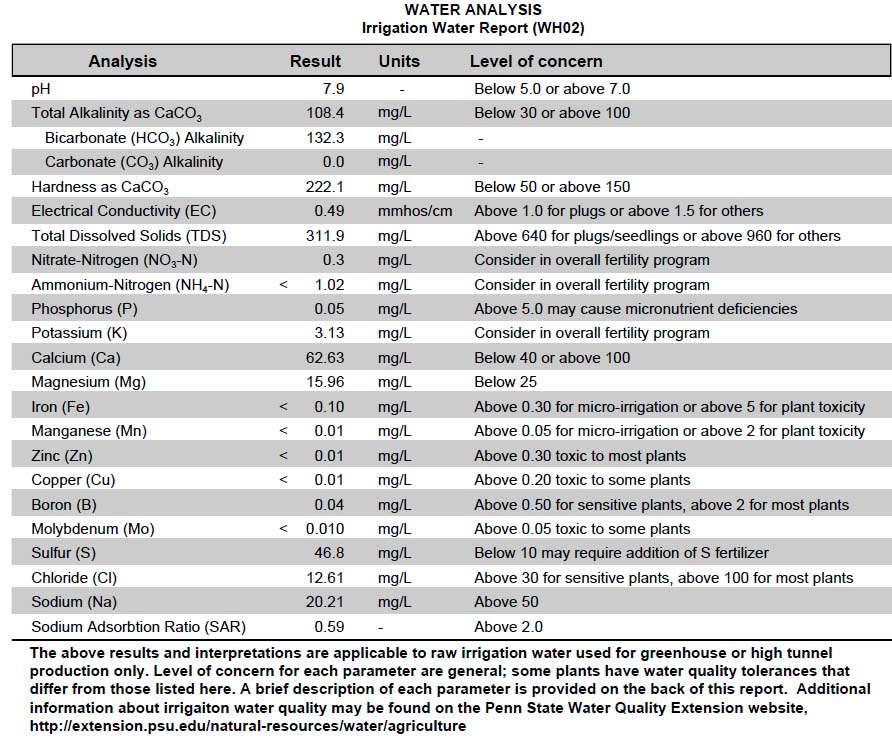
3. Order a Water Test: Collect a water sample according to directions and submit to a qualified water analysis laboratory. Universities are a great resource for this, as many have agriculture extension programs that work exclusively with farmers. They will also provide irrigation water analysis, which will contain much more relevant information than a drinking water analysis. This is the most accurate method to determine the quality of your water and what factors may affect plant health and performance. See below for an example of a water analysis report.
WATER QUALITY ANALYSIS
The following report from Penn State Water Quality Extension shows high pH, bicarbonate alkalinity and hardness. Alkalinity has a direct correlation with elevated soil pH values and reduced micronutrient uptake. Alkalinity in water should not be confused with an alkaline, or basic, pH. In terms of water quality analysis, alkalinity refers to the content of bicarbonates or carbonates in the water and the buffering capacity that provides.
When bicarbonates or carbonates are in excess, they form insoluble complexes from magnesium and calcium, causing sodium to become the dominant ion in the water. This leads to a lack of available calcium and magnesium to the plant and interferes with water uptake by the roots. As the plant removes minerals from the solution and as moisture evaporates, those insoluble complexes can also accumulate in soil and clog irrigation equipment. High pH is also more likely to accumulate soluble salts in the growing medium, further interfering with nutrient uptake and causing unpredictable pH fluctuations.
WATER QUALITY IMPROVEMENTS
Adjust Alkalinity: There are several acids available for horticultural use that are an effective tool to reduce alkalinity of irrigation water.
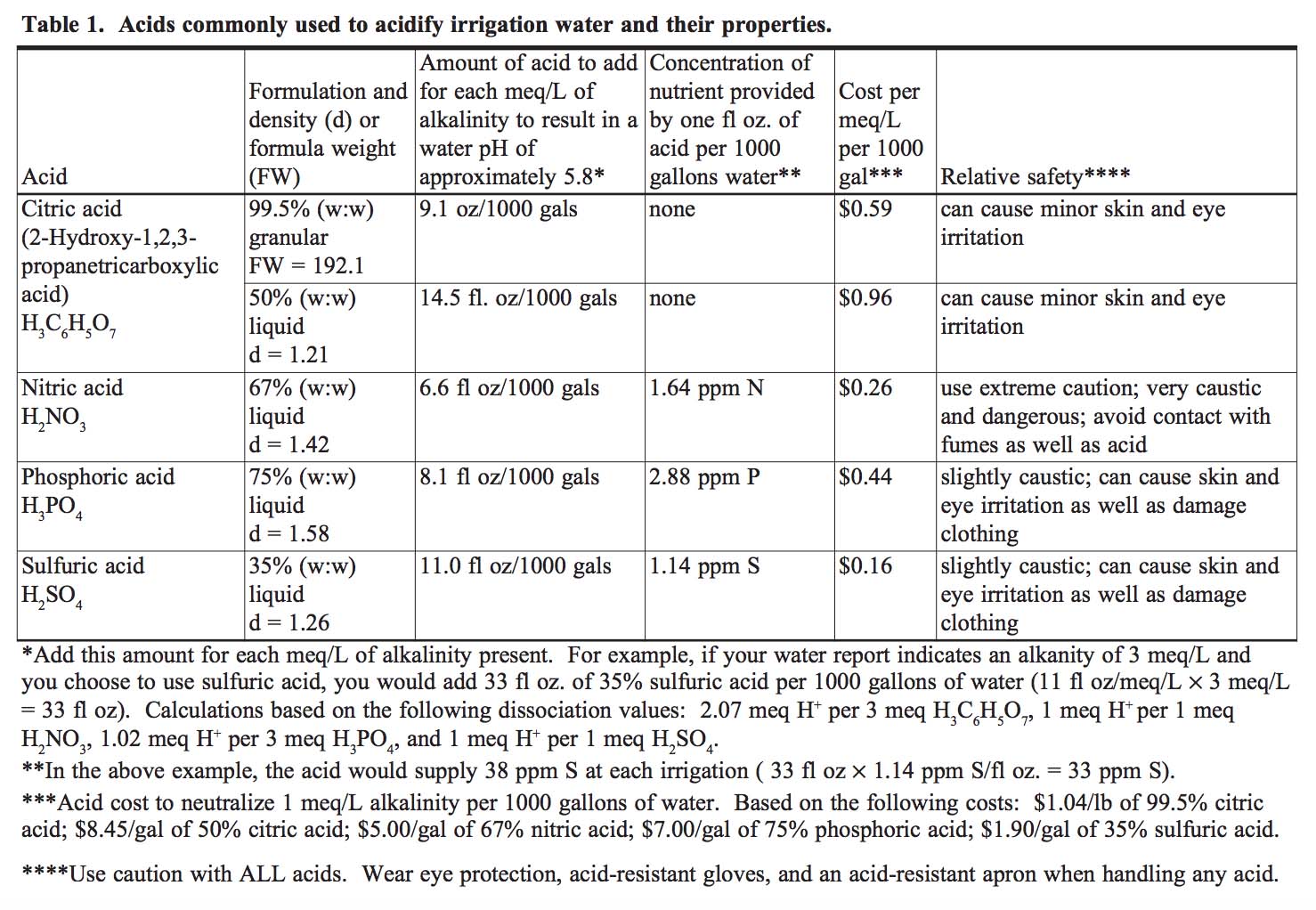
Filtration Systems: If there are known pathogens in the water supply, there are several options available to reduce contamination. Chlorine is commonly used in drinking water supplies, but high residuals may have a negative effect on plant health. If high salts or heavy metals are a problem, reverse osmosis filtration is the best solution to produce pure water. Other inexpensive alternatives include acid injections prior to irrigation to neutralize alkalinity and ultraviolet filters to kill unwanted organisms in the water. Table 2 outlines filtration options for various water quality issues.
Table 2. Filtration Options
| Total Dissolved Solids | Bicarbonate & Carbonate | Calcium & Magnesium | |
|---|---|---|---|
| Reverse Osmosis | X | X | X |
| Deionization | X | X | X |
| Anion Exchange | X | ||
| Water Softening (cation exchange) | X | ||
| Acid Injections | X |
CONCLUSION
Aside from pest management, water quality analysis is one of the most important preventative and diagnostic tools available to growers. Prior to using water for irrigation, obtain a detailed analysis from a qualified water testing lab to identify and correct any potential future problems. If plant health appears to be compromised, water quality should always be the first item to check off the troubleshooting list.
Poor water quality can interfere with nutrient availability and uptake, worker health, and plant performance and yield. Paying attention to water quality and taking necessary steps to correct issues will prolong equipment operation, prevent crop losses and ultimately save the grower money and contribute to continued success.
APPENDIX A
Information Resources
Table 1: https://www.ces.ncsu.edu/depts/hort/floriculture/plugs/alkalinity.pdf
Acid adjustment calculator: http://extension.unh.edu/Agric/AGGHFL/alk_calc.cfm http://www.cropnutrition.com/nutrient-knowledge
APPENDIX B
| EXCESS ELEMENT | NUTRIENT(S) AFFECTED |
|---|---|
| Nitrogen | Potassium, Calcium |
| Potassium | Nitrogen, Calcium, Magnesium |
| Phosphorus | Zinc, Iron, Copper |
| Calcium | Boron, Magnesium, Phosphorus |
| Magnesium | Calcium, Potassium |
| Iron | Manganese |
| Manganese | Iron, Molybdenum, Magnesium |
| Copper | Molybdenum, Iron, Manganese, Zinc |
| Zinc | Iron, Manganese |
| Molybdenum | Copper, Iron |
| Sodium | Potassium, Calcium, Magnesium |
| Aluminum | Phosphorus |
| Ammonium Ion | Calcium, Copper |
| Sulfur | Molybdenum |






